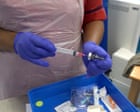
Thousands of cancer patients across England are about to experience a profound shift in how they receive life-saving treatment. The National Health Service is rolling out a seven-minute immunotherapy injection that promises to replace hours of intravenous infusions. This advancement centers on the drug atezolizumab, manufactured by Roche. For patients battling a range of cancers, including lung, breast, and liver disease, the change means less time in hospital beds and a significantly reduced burden on overstretched oncology clinics.
Yet, the announcement raises urgent questions regarding the logistical hurdles of implementation, supply chain resilience, and the true cost of expedited medical innovation within a struggling public healthcare system. Behind the optimistic press releases lies a complex reality of adoption, infrastructure readiness, and patient care. Expanding on this theme, you can also read: The Seven Minute Cancer Jab Is a Logistics Mirage.
The Science Behind the Seven Minute Shift
Immunotherapy has fundamentally changed oncology. However, the physical reality of receiving these treatments remains grueling for patients. Standard intravenous infusions of atezolizumab often take anywhere from thirty minutes to an hour to administer, requiring continuous clinical supervision and dedicated infusion chair space.
The new formulation uses a subcutaneous injection. Atezolizumab is combined with recombinant human hyaluronidase, an enzyme that breaks down the hyaluronic acid in the extracellular matrix beneath the skin. This enzymatic breakdown allows the drug to disperse rapidly, enabling the delivery of a substantial dose in a fraction of the time. Observers at WebMD have also weighed in on this matter.
Clinical trials, including the IMscin001 study, demonstrated that pharmacokinetic exposure and efficacy are comparable to the intravenous route. The safety profiles remain largely consistent, with injection site reactions being the primary new consideration.
But science does not exist in a vacuum. A faster administration time shifts the bottleneck from the infusion chair to the pharmacy and the supply chain. Ensuring that every regional NHS trust can store and handle the new formulation requires precise temperature controls and trained personnel.
The Economic and Clinical Trade offs
The introduction of subcutaneous atezolizumab is not purely an act of altruism; it is a strategic maneuver in pharmaceutical lifecycle management and healthcare optimization. The economic implications are vast.
By reducing the time spent in the clinical setting, the NHS stands to save millions in staff hours and facility overhead. Nurses who previously spent hours monitoring IV lines can pivot to more complex clinical tasks or patient support.
Yet, we must address the financial reality of newer formulations. Pharmaceutical companies frequently seek new patents and extended exclusivity periods for subcutaneous delivery methods. This practice can delay the introduction of cheaper biosimilars, potentially keeping costs higher for healthcare systems over the long term.
Furthermore, switching thousands of patients requires an extensive retraining program for oncology nurses. If an NHS trust lacks the resources to implement this training, the benefits will remain confined to well-funded metropolitan centers, exacerbating existing healthcare inequalities.
Navigating the Logistical Choke points
The deployment of a drug delivery system is only as strong as its weakest link. For the National Health Service, the current infrastructure is under immense strain. Waiting lists for cancer diagnostics and treatments remain at historic highs.
Hospitals must now re-evaluate their pharmacy dispensing workflows. Subcutaneous atezolizumab is supplied as a ready-to-use liquid in a vial. While this eliminates the need for reconstitution, it places a higher demand on cold-chain storage and inventory management.
Hospitals that experience power fluctuations or lack adequate refrigeration units risk wasting highly expensive biologics. The administrative burden of tracking patient doses, managing expiration dates, and coordinating with regional suppliers requires robust digital infrastructure, which many NHS trusts still lack.
The Patient Experience and the Limits of Speed
For a patient, a seven-minute injection versus an hour-long infusion is more than just a time-saving metric. It represents a psychological reprieve. Sitting in an infusion suite serves as a stark, continuous reminder of illness. Shorter appointments allow patients to return to their normal lives faster, reducing hospital-induced anxiety.
However, we must confront the reality that speed does not equal a cure. Atezolizumab is an immune checkpoint inhibitor, and it carries significant risks of immune-related adverse events. Patients receiving the injection still require close monitoring for side effects such as pneumonitis, hepatitis, and severe endocrinopathies.
The physical speed of administration does not alter the biological timeline of the treatment's effect. The body still requires time to recognize and attack the cancer cells. Overemphasizing the speed of the injection risks trivializing the ongoing, grueling reality of systemic cancer therapies.
The Broader Implications for Global Oncology
What happens in England will serve as a blueprint for the rest of the world. Healthcare systems across the globe are watching to see if the subcutaneous rollout reduces the backlog of cancer treatments.
If the NHS succeeds in transitioning thousands of patients, regulators in the United States and the European Union will likely fast-track similar approvals for other monoclonal antibodies and immunotherapies. This shift could signal the beginning of the end for the traditional, chair-bound infusion center as the primary model for biologic delivery.
However, the transition demands transparency from regulators and manufacturers regarding pricing and access. Without clear guidelines on how savings are reinvested into front-line patient care, this development risks becoming a triumph of logistics rather than a genuine leap forward in cancer outcomes.
The true test of this innovation lies in the coming months as the NHS attempts to scale the rollout across dozens of regional trusts. The infrastructure will be tested, the supply chains will be stretched, and the resilience of the system will be laid bare.